3Patti Games APK Download
More »










Popular Apps

S9 Pro GameS9 Pro Game Inc

Teen Patti EliteTeen Patti Elite Inc

3 Patti Land3 Patti Land Inc

Teen Patti RealTeen Patti Real Inc

3 Patti Best Bet3Patti Best Bet Inc

3 Patti Ludo3 Patti Ludo Inc

3Patti Flying ChessFlying Chess Inc

Teen Patti BestTeen Patti Best Inc

Teen Patti MaxTeen Patti Max Inc

Slots 777Slots 777 Inc
More »










Editor's Choice

3 Patti Master3 Patti Master Inc

Teen Patti RealTeen Patti Real Inc

Teen Patti BestTeen Patti Best Inc

Card RummyCard Rummy Inc

3 Patti Loot3 Patti Loot Inc

Teen Patti ShowyTeen Patti Showy Inc

Teen Patti SpinTeen Patti Spin Inc

Teen Patti Happy ClubTeen Patti Happy Club Inc

Teen Patti MelaTeen Patti Mela Inc

S9 Pro GameS9 Pro Game Inc
3Patti Games Pakistan Download
Download the latest version of 3 Patti APK to enjoy multiple games and make real money. Claim your free rewards and bonuses for free. Free download the Latest version of 3 Patti APK - Tiger Dragon plays to earn real money from different games. Download 3 Patti Pakistan Apk for your Android device for free. Play various games like Dragon vs Tiger, Minies, and many more to earn money online. Download and Play TeenPatti Cash, classic Poker online anytime, anywhere. Play teen patti, 7updown, 3 patti, slot machine game, crash. Download 3 Patti Pakistan APK file latest official version for Android from this page and play many bet games & earn money!Download Teen Patti Games APK
Teen Patti games are mobile app that lets you play the popular card game Teen Patti (also known as 3 Patti) against real people online. Download Trusted Teen Patti games like Teen Patti Blue, Gold, Vegas, Lucky, Loot, Room, Sky, World, Master and many Teen Patti Games. Teen Patti Game Online Earning platform where you can download various rummy application such as Teen Patti Master, Teenpatti Gold, Taurus Cash. Teen Patti APK is Android-based real cash earning app that consists of multiple games. Invest money on the available games to generate revenue.3 Patti Games Latest Version
3 Patti games are legitimate online earning apps where people from different areas join to play the game and earn real money living in Pakistan. Download 3 Patti Games Pakistan Official APK and earn money playing betting games such as Teen Patti, Rummy, Car Roulette, and many others. Free download the latest version of Teen Patti Sky (3 Patti) Games on Android. Join now to earn real cash and withdraw to EasyPaisa and Jazzcash. The new app allows you to play more than 30 free games and can invest a minimum of 100. This apps contains all the latest casino games. Now you will be able to play any game of your choice.Teen Patti APK for Android
Download and install the Teen Patti Games APK on your Android device. Then, invite your friends and family to play games online or simply start a challenge. Teen Patti games are earning apps that allow you to play and earn money from the comfort of your home. You can easily download these apps and start earning real cash without any hassle. Play various games like Dragon vs Tiger, Minies, 7Up Down, Fruitline, and many more to earn money online. Teen Patti Games offers many games to play and make real cash through multiple ways. Login is the first step towards safeguarding your personal information and gaining access to a wide range of features.Download Install 3Patti Games
Follow These Steps:
- Visit Our Website: Go to our official website and navigate to the Teen Patti Games section.
- Download the APK: Click the Download Now button to get the latest version of the Teen Patti game APK file.
- Enable Unknown Sources:
- Open your phone's Settings.
- Go to Security or Privacy.
- Enable Allow Installation from Unknown Sources.
- Install the Game: Open the downloaded APK file and tap Install. Wait for the installation process to complete.
- Launch and Enjoy: Open the app, sign in or create an account, and start playing Teen Patti games to earn money from home!




 3Patti World Download
3Patti World Download
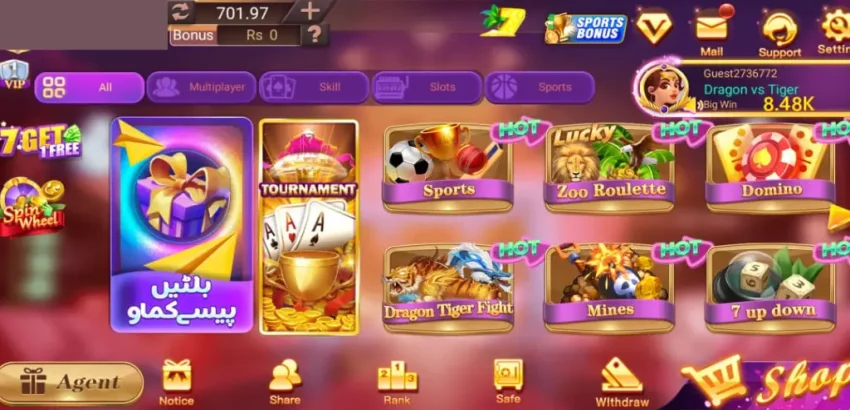
 3Patti Sky Download
3Patti Sky Download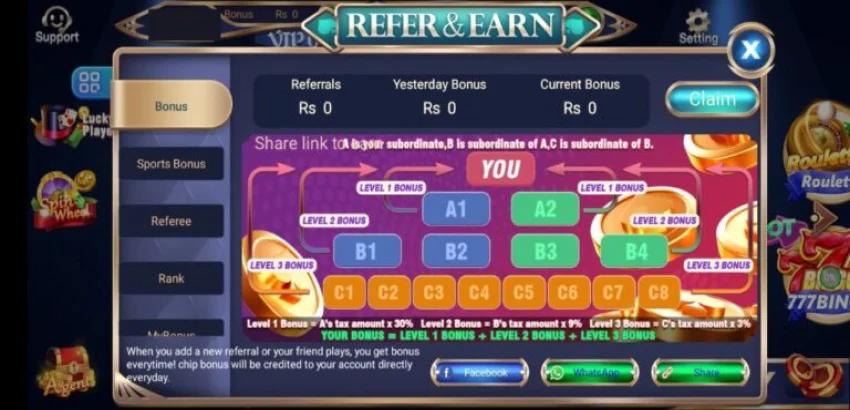
 3Patti Room Download
3Patti Room Download
 TeenPatti Gold Download
TeenPatti Gold Download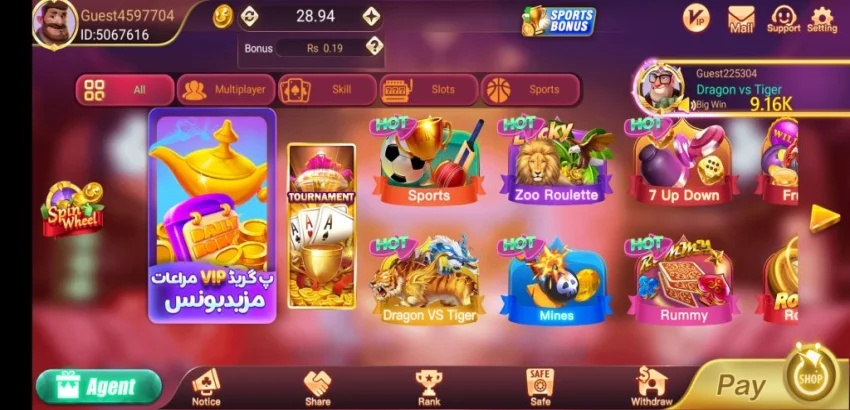
 3Patti Lucky Download
3Patti Lucky Download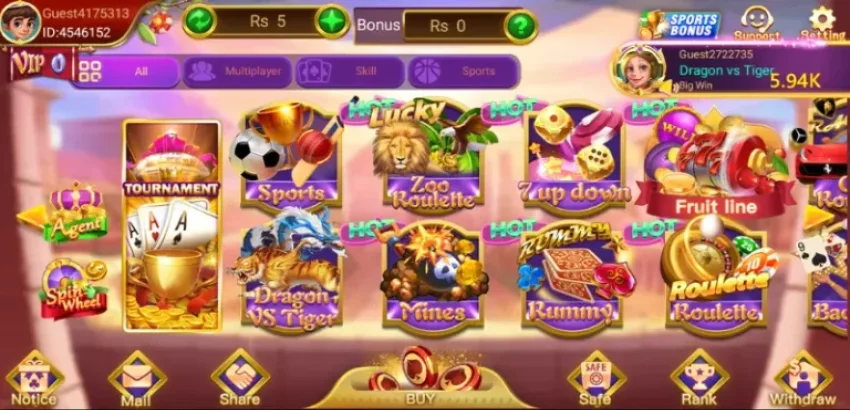
 3Patti Blue Download
3Patti Blue Download
















